Systemic Lupus Erythematosus: Personalized Care to Improve and Resolve Symptoms
Because it can affect so many different parts of the body, it can be difficult to diagnose and manage. It can also have more serious effects on health, such as when the blood vessels are involved, something I illustrate in my patient case below.
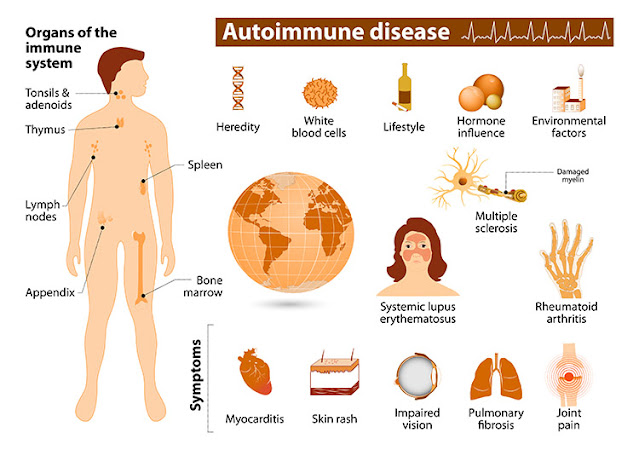
Lupus is more formally known as systemic lupus erythematosus (SLE). Although researchers have identified multiple types of lupus, the truth is that lupus manifests in different ways for different people.Because lupus is an autoimmune attack against DNA, the disease can manifest in the joints, the brain, the skin, the organs, or other tissues. That’s what makes it so difficult to diagnose.
For instance, someone with undiagnosed lupus and joint pain may see a rheumatologist, but tests for rheumatoid arthritis come back negative.
Or they may have elevated kidney markers, but the labs aren’t bad enough for further testing, and they are put on the “wait-and-see” approach.
Another may have mysterious neurological symptoms, and another chronic skin rashes or eczema-type symptoms. But rarely is lupus investigated.
While the standard health care model looks for organ failure or a butterfly rash on the face to screen for lupus, these symptoms may never appear for many patients. The hallmark butterfly rash can appear on someone’s leg or arm. That’s why it can take decades for many lupus patients to receive a diagnosis.
The initial screening for lupus is an anti-nuclear antibody (ANA) test. It’s important to understand that sometimes antibody tests can come back negative as immune function fluctuates, and a second test may be needed. A more complete lupus panel that screens for antibodies against many different DNA fragments confirms a lupus diagnosis in the standard health care model.
When lupus affects the nervous system and brain, it can cause brain fog, depression, memory problems, behavioral changes, confusion, migraines, and strokes.
When the cardiovascular system is affected, such as in my patient’s case below, signs can include hardening of the arteries, coronary artery disease, and heart attack.
The skin is another target with rashes, sores, and ulcers.
When supporting those suffering from an autoimmune disease such as lupus, it doesn’t matter which tissue is being attacked—the primary strategy is to find the triggers for inflammation and autoimmune flare-ups and remove or mitigate those.
Most autoimmune patients must also follow an anti-inflammatory diet and lifestyle to stabilize immune function and promote the dampening of inflammation.
Autoimmune disease is now more common than ever, although statistics show that women are disproportionately affected, being twice as likely to be diagnosed.
Other common autoimmune diseases include Hashimoto’s thyroiditis, multiple sclerosis, rheumatoid arthritis, psoriasis, vitiligo, celiac disease, Graves’ disease, Type 1 diabetes, lupus, and some inflammatory bowel diseases. To learn more about autoimmunity, please read my other articles in The Epoch Times.
Marta’s Story: Lupus and the Cardiovascular System
I see patients with complex and chronic autoimmune and neurological disorders. Because Marta’s lupus affected her cardiovascular system, she was one of the more critical.
If you look at Marta, she looks very healthy. No one would look at her and think she suffers from a severe case of lupus.
However, her swelling is so bad that socks are too binding for her ankles and hurt her toes. She can’t handle the feeling of blankets on her body. She wakes up stiff, aching, and with brain fog. Her vision is blurred, and she has chronic migraines and chest pains.
Marta had a heart attack three days after her 40th birthday. It’s believed lupus caused her heart attack because when the surgeon tried to place a stent in her artery, the artery tore, and the surgery was unsuccessful. She was diagnosed with lupus shortly after.
Autoimmune disease doesn’t manifest overnight. It often progresses over years, destroying tissue until the symptoms become severe enough for a medical diagnosis. You don’t get a heart attack and a dissecting artery in surgery overnight. Instead, it results from long-term inflammation and damage to the artery.
Marta’s symptoms began in 2000 with numbness and tingling on the left side of her face, for which she was prescribed gabapentin, an anticonvulsant medication used to treat nerve pain, for which swelling of the hands and feet is also a common side effect, further complicating a future diagnosis.
Her doctor didn’t investigate the possibility of neurological autoimmunity.
Her heart attack occurred a year later, leaving her with chest pains, shortness of breath, weight gain, swelling throughout her body, and feeling pressure from the extreme systemic inflammation.
Marta’s symptoms continued to snowball. She developed back pain, joint pain, and early spinal degeneration, and she bruised easily. These symptoms could indicate joint autoimmunity, and easy bruising is commonly a symptom of anti-phospholipid autoimmunity, which can occur as a primary condition or in the setting of lupus.
By the time she came to see me, everything was hurting. She had migraines despite taking migraine medication. She was also in cardiac rehabilitation three times a week.
She also had developed some concerning kidney symptoms and markers suggesting kidney infection or kidney disease related to lupus.
In Marta, we see inflammation and autoimmunity spreading throughout her body, resulting in an ever-increasing array of doctors and medications. This isn’t uncommon in lupus patients.
Her condition puts her at high risk for both a stroke and another heart attack and illustrates how severe autoimmunity can be once the vascular system is involved.
Although her exam intake showed signs of neuroinflammation, such as brain fog, fatigue, and poor memory, focus, and concentration, thankfully, a brain scan showed no white matter lesions that indicate advanced brain degeneration. Lupus increases the risk of brain inflammation and degeneration and can lead to scarring and injury of pathways in the brain and spinal cord. In Marta’s case, this is especially a concern given her vascular issues, which may deprive the brain of healthy circulation.
Marta also tested positive for an autoimmune mixed connective tissue disease (MCTD). The antibody for this disease, anti-U1 ribonucleoprotein (RNP), is often present in people with both MCTD and lupus.
Lupus and a connective tissue disease make it more likely to see things such as retinal detachment, an injury to the connective tissue, and vasculature of her eye. In fact, Marta was working with an ophthalmologist for blurred vision, double vision, and retinal detachment.
MCTD also affects vascular tissue and joints, likely playing a role in the signs and symptoms in those areas.
Before her heart attack, Marta took no medications and didn’t even like to take Tylenol. But because she developed so many symptoms, she now works with multiple doctors and is on multiple medications.
Her daily list of medications includes a beta blocker, statins, a blood thinner, anti-inflammatories, prednisone, nitroglycerine, a migraine medication, and gabapentin, to name a few. Because she develops infections easily, she also has been given antibiotics on several occasions. She understandably worries about side effects and the long-term effects.
This is a common scenario with more severe autoimmune cases. With lupus, medications are necessary during an inflammatory crisis to prevent permanent tissue damage.
In Marta’s case, her treatment could help prevent another heart attack or blindness. Marta was lucky to receive good care from her team of doctors: a cardiologist, rheumatologist, ophthalmologist, nephrologist, neurologist, and general physician. Her medical team monitors injury to specific organs and tissues and prescribes anti-inflammatory and immune suppressive medications as needed.
However, this model doesn’t identify lifestyle, environmental, or dietary factors that may play an underlying role in triggering her immune response.
When a patient with autoimmunity fails to manage the underlying autoimmune mechanisms, the condition worsens as the immune system continues its deranged attack on the body. The average patient goes from one doctor to another, trying to figure out why things are getting worse instead of better and receiving one prescription after another.
The personalized lifestyle medicine model of managing autoimmunity isn’t part of the standard medical or insurance model. This model involves identifying immune triggers—foods, chemicals, infections, and lifestyle stressors—and guiding the patient in making dietary and lifestyle changes to dampen inflammation and put autoimmunity into remission.
Lowering inflammation improves cellular health so that the body is better able to regulate autoimmunity. Daily physical activity, even in pain, is vital for most patients. For instance, studies show that patients with advanced and painful rheumatoid arthritis experience reduced pain and autoimmune expression with daily exercise. These anti-inflammatory dietary and lifestyle changes activate immune cells that combat autoimmunity and shift the body into a recovery state.
Marta’s Personalized Lupus Protocol
An autoimmune diet typically involves following the elimination-provocation diet for four to six weeks. This diet eliminates most foods other than meats and non-starchy vegetables and can help a patient identify which foods are inflammatory triggers.
For Marta, this was phase one of her protocol. Once inflammation comes down, I would instruct her to introduce daily, low-impact exercise, such as using a stationary bike or walking.
Once those habits were established, and inflammation continued to come down, I’d implement customized nutritional, cardiovascular, and insulin management support based on her lab tests.
I would also test for immune reactions to specific foods, environmental chemicals, and pathogens to determine if these were triggering her autoimmunity.
For instance, Marta worked on a farm growing up, which exposed her to multiple toxic chemicals. She also has breast implants, a foreign material introduced to her immune system that may trigger an immune reaction, particularly with leaking implants.
Chemicals such as pesticides, benzene in car exhaust or cigarette smoke, and BPA in plastics can trigger and exacerbate autoimmune responses.
We can screen for this by checking for immune antibodies made in response to these chemicals. In other words, the immune system responds to the chemicals as if it is an immune trigger such as gluten.
This can be helpful information when it isn’t obvious what triggers are flaring up the autoimmune expression. The amount of chemicals in your body in this scenario isn’t as important as whether you are reacting to them.
I would also want to evaluate her microbiome and gut integrity.
These strategies are also diagnostic. Will going off gluten reduce migraines and joint pain? Marta wasn’t on a gluten-free diet, something I find helps most of my autoimmune patients. Gluten can be a profound trigger of autoimmune destruction and symptoms.
Are specific foods or chemicals connected to specific symptoms? Do any of the strategies banish the brain fog?
What role did infections play? Marta’s symptoms worsened when she developed COVID-19. It’s common for viruses, including COVID, to flare up autoimmunity. I co-authored a study that found 28 different autoimmune responses can be triggered by the SARS-CoV-2 spike protein.
Marta has had many throat infections, pneumonia twice, and inflammation in the chest, all of which are triggers.
None of this suggests that a personalized diet and lifestyle plan should replace her conventional treatment. With a condition as serious as lupus, she still needs conventional treatment to reduce her inflammatory load and prevent disease progression. We also have to evaluate and monitor any interactions between her medications and nutraceutical supplements.
This is all part of managing a very progressed and complex case of systemic lupus. Other autoimmune diseases, such as Hashimoto’s hypothyroidism or Type 2 diabetes, also require medical management with prescription drugs.
However, regardless of the tissue being attacked or the conventional treatment required, managing the underlying autoimmune mechanisms cannot be overemphasized. Autoimmune management can significantly improve the patient’s quality of life and slow or halt the progression of autoimmunity, including to neurological tissue.
A Not-So-Happy Ending
I wish I had a happy clinical ending to Marta’s story. She was distressed about her condition and the many medications she was on when she first came to see me.
However, despite the gravity of her condition, Marta found the changes required to stop the autoimmune progression were more than she could handle.
She was part of a large family that gathered regularly, and the foods they ate were an integral part of the gatherings. Her family didn’t understand that gluten, dairy, and corn prevented her from slowing or stopping the progression of her disease and relieving her symptoms.
Marta’s symptoms also indicated brain inflammation, which commonly causes depression. It’s possible that Marta was simply too depressed to handle eating differently than her family.
Thankfully, most of my patients can make the necessary changes to manage their autoimmunity, especially once they experience how much better they feel. But not all are. Sometimes, this includes the parents of a child or teen with significant autoimmune issues who don’t want to deprive their child of a “normal” life.
These are the paths both patients and practitioners must navigate in a preventive model. Patients must educate themselves, make drastic changes, and assert themselves with loved ones and in social situations.
But many say their autoimmune disease helped them grow, find more balance, and prioritize what is most important in life.
About the Author: Datis Kharrazian, Ph.D., DHSc, DC, MS, MMSc, FACN, is a Harvard Medical School-trained, award-winning clinical research scientist, academic professor, and world-renowned functional medicine healthcare provider. He develops patient and practitioner education and resources in the areas of autoimmune, neurological, and unidentified chronic diseases using non-pharmaceutical applications.
Reposted from: https://www.theepochtimes.com/health/lupus-complex-confusing-and-difficult-to-treat_4872659.html


.png)





.png)

Comments
Post a Comment