Bioshield Shows Sustained Early Promise in Recurrent GBM: Initial 100% Disease Control in Pilot Cohort, with Ongoing Trials - Dr. Patrick Soon-Shiong
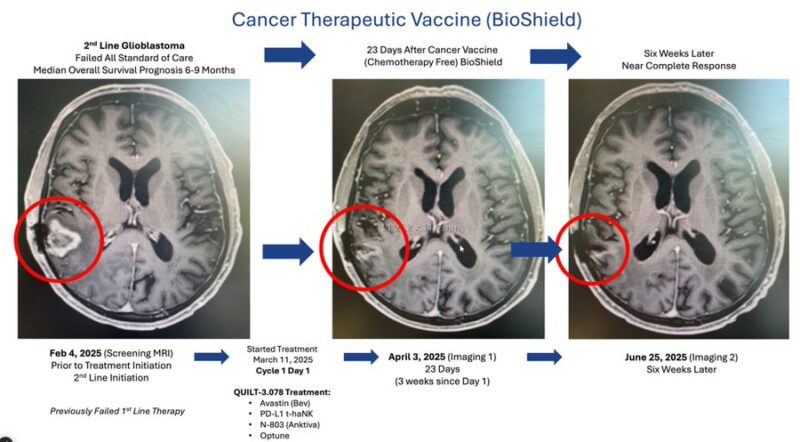
In a series of posts on X, the ImmunityBio executive chairman called glioblastoma “medicine’s biggest challenge,” pointing to decades of limited progress with surgery, radiation and chemotherapy. Standard regimens, he noted, in 90% of cases deplete lymphocytes, crippling NK and T cells and leaving patients prone to relapse.
The new approach—described as a “chemo-free” combination of Bioshield (Cancer Vaccine*), NK cell therapy plus an immune-stimulating device—aims to protect and rescue the immune system rather than destroy it.
So far, five patients with recurrent GBM have been treated. According to Soon-Shiong, two patients have achieved near-complete responses, while all five have shown 100% disease control. “When the immune system is protected, CR in tumors difficult to treat in recurrent GBM is possible,” he wrote.
Asked about the other three patients, Soon-Shiong said that one additional responder was seen early in the trial, and that the remaining two patients have just begun therapy and remain stable as they await follow-up MRIs.
The data are preliminary and based on five patients, but Soon-Shiong signaled that more readouts are on the way. ImmunityBio expects to present lung cancer data at the World Conference on Lung Cancer (WCLC25) next year.
The glioblastoma trial is open at CSSIFM.org.
Recent Platform Validation: Durable Complete Remission in Waldenström's Macroglobulinemia (January 2026)
On January 16, 2026, Dr. Soon-Shiong shared results (X.com) from a compassionate-use case in relapsed/refractory Waldenström's macroglobulinemia:What's Your ALC?: ImmunityBio's Dr. Pat Soon-Shiong Details Lymphopenia Dangers and Anktiva's Cancer Treatment Role
Our NK snd T cells have a highly specialized receptor on their surface called IL15. Ankitiva is a protein we have manufactured that is given as a subcutaneous injection and binds to this IL 15 receptor on the surface of these cancer killing cells, and when this happens these protective and cancer killing NK and T cells expand in huge numbers and become activated. That’s the Bioshield. That’s what is needed to kill cancer and to drive what we call Memory T cells. And we measure the levels in your body of these NK and T cells by a simple blood test called ALC. The Absolute Lymphocyte Count.
ImmunityBio Company Review (2026)
ImmunityBio, Inc. (NASDAQ: IBRX) is a vertically integrated, clinical-stage biotechnology company headquartered in Culver City, California, specializing in targeted immunotherapies and cell therapies designed to enhance the human immune system against cancer and infectious diseases. Founded by Dr. Patrick Soon-Shiong, a surgeon and inventor known for developing ABRAXANE (sold to Celgene in 2010), the company emphasizes "Smart Therapies for Difficult Diseases®," focusing on activating natural killer (NK) cells, T cells, and immunological memory to achieve durable responses without severely weakening the immune system, unlike traditional chemotherapy or radiation. Their approach, dubbed "Immunotherapy 2.0" or "Cancer BioShield™," aims to treat the host rather than just the tumor, with platforms including cytokine fusion proteins, vaccine vectors, and engineered NK cells. As of March 2026, ImmunityBio employs around 600 people and operates with a market cap of approximately $8 billion, though exact figures fluctuate with stock volatility.Products and Pipeline
- Recurrent Glioblastoma: Early 100% disease control (n=5); randomized trials starting.
- Waldenström's macroglobulinemia: Durable complete remission (15+ months ongoing) in relapsed/refractory patient using off-the-shelf CAR-NK + Rituximab.
- NSCLC: Positive lymphopenia reversal with checkpoint inhibitors; Saudi approval for metastatic combo.
- Bladder: Strong commercial traction; EU conditional authorization progressing.
- Other: Long COVID trial, prostate combos, CAR-NK platforms.
QUILT trials
A "QUILT trial" refers to a series of clinical studies, primarily by ImmunityBio, testing novel immunotherapies (like N-803 (nogapendekin alfa)/ANKTIVA) combined with existing treatments (e.g., BCG, checkpoint inhibitors) for various advanced cancers, especially non-muscle invasive bladder cancer (NMIBC) and non-small cell lung cancer (NSCLC). These trials aim to boost anti-tumor immunity, converting non-responders or those with stable disease into responders, showing promising durable complete responses in difficult-to-treat cancers.Key QUILT Trials & Focus Areas:
- QUILT-3.032 & QUILT-2.005: Focus on Non-Muscle Invasive Bladder Cancer (NMIBC), using N-803 (ANKTIVA) with BCG, showing high complete response rates in both BCG-naive and BCG-unresponsive patients, with long-lasting remissions.
- QUILT-3.055: Investigates combination immunotherapies (N-803 plus checkpoint inhibitors) in advanced solid tumors, including NSCLC, for patients who have progressed on prior PD-1/PD-L1 therapy.
- Other QUILT Studies: Also explore therapies for pancreatic cancer and other cancers, aiming to activate immune cells (NK cells, T cells) for durable responses.
- Bladder Cancer: High complete response rates (e.g., 71% in BCG-unresponsive NMIBC) with long durations and high rates of avoiding cystectomy (bladder removal).
- NSCLC: Converts patients with stable disease on immunotherapy to responsive disease with the addition of N-803, showing promising overall survival.
Financial Performance
Employee and Culture Reviews
Employee sentiment is mixed to negative. On Glassdoor, only 23% recommend working there, with a 3.0/5 work-life balance rating; complaints include limited career growth and high-pressure environment. Indeed reviews echo this: "Poor work environment" and no advancement opportunities, though some praise hands-on experience in manufacturing. Culture scores low, with reports of delays in promotions and a demanding biotech pace.Public and Market Sentiment
On X (formerly Twitter), sentiment leans positive toward scientific breakthroughs: Users hail ANKTIVA/BioShield for potential in glioblastoma, Long COVID, and "turbo cancers," urging FDA approvals and amendments to "Right to Try" for frontline use. Calls tag figures like @RobertKennedyJr and @DrMakaryFDA for faster action. Investor frustration focuses on stock manipulation by shorts and big pharma, with pleas for BioShield to combat viruses/cancers. Some compare it favorably to competitors like Northwest Biotherapeutics. Broader reviews praise innovation but note risks: High cash burn, manufacturing disruptions, and dependency on trial success. Analyst views: Promising upside but biotech volatility; one calls it a "high-growth tech stock" analog.Strengths and Weaknesses
Strengths: - Innovative platforms with real-world impact (e.g., ANKTIVA approvals, glioblastoma data). - Revenue ramp-up and regulatory wins signal commercialization strength. - Broad applicability (cancer prevention, viruses) positions it as a leader in immunotherapy. - Strong leadership and collaborations (NCI, VA hospitals). Weaknesses: - Financial strain: Cash burn, dilution from financings, short-selling pressure. - Employee morale issues could impact retention/innovation. - Pipeline risks: Many therapies investigational; delays in BLA submissions (e.g., NMIBC papillary). - Competition from established players (e.g., Keytruda, CAR-T) in crowded immunotherapy space.- Multiply.
- Stay alive.
- Become more cytotoxic.
Used thoughtfully, Anktiva can:
Expand the pool of effector cells.
Increase their killing capacity.
Enhance antibody‑dependent killing (for example alongside anti‑PD‑L1 or HER2‑targeting antibodies).
In non‑muscle‑invasive bladder cancer, the logic is particularly good:
BCG in the bladder already creates a highly inflamed, immune‑rich environment.
There are plenty of T‑cells and NK cells in and around the tumour.
Anktiva then adds fuel to this existing immune fire, and clinical responses reflect that synergy.
In inflamed melanoma, or potentially inflamed regions of other cancers with strong immune presence, the same logic applies. Take off the PD‑1 brakes, add IL‑15‑style fuel, and you can see deeper and more durable responses – at least in a subset of patients.
Where I start to push back against the hype is when Anktiva is presented as a universal solution that somehow overcomes any microenvironment.
Anktiva does not:
Dissolve collagen.
Neutralise CXCL12 barriers on its own.
Switch off TGF‑β or IL‑6‑rich CAFs.
Magically create T‑cells in severely lymphopenic or heavily pre‑treated patients.
If the stroma is acting like concrete and the tumour is immunologically “cold,” Anktiva is, at best, a strong cheerleader shouting from outside the stadium.
I’m not against Anktiva. I’m against context‑free enthusiasm.
The future of immune‑oncology (IO), as I see it, belongs to people who:
Respect the importance of stroma and microenvironment.
Use local primers and metabolic strategies to create antigen, danger and access.
Then, only when the roads are opening, consider carefully how much fuel to pour on – and for whom it really makes sense.


.png)







Comments
Post a Comment