Information related to the COVID-19 ('CO' stands for corona, 'VI' for virus, and 'D' for disease) pandemic has been overwhelming and confusing as well. Information is all over the place and various groups are giving conflicting statements. How do you make sense from all these fragmented information?
As of August 2021, there are more than 6,000 studies that have been launched to investigate various treatments for COVID-19. You can review the details of these trials on ClinicalTrials.gov. New ones are being added every day. The aim of this article is to organise and summarise relevant information in one place. Below, we look at the most studied categories.
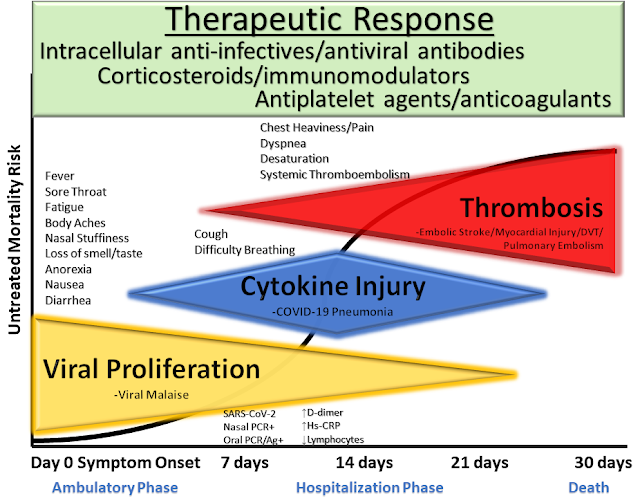 |
| McCullough et al. Reviews in Cardiovascular Medicine, 2020 |
Most doctors know the need to focus now on early treatment as the most immediate and practical way to reduce hospitalisations and death. This is your guide to help you know your options, and to use with your personal physician.
Below, we look at the best outpatient treatment categories for COVID-19 and summaries of the rationale and evidence for each category.
1. Vaccine
Vaccine is a preventive strategy to boost your immune system and reduce the risk of getting COVID-19. Therefore technically, vaccine is not considered a treatment but rather a preventive strategy.
As of August 2021, researchers are currently testing more than 90 vaccines in clinical trials on humans. The vaccine remains a popular agent for COVID-19 protection, and the published reports of the candidate vaccines showcase some encouraging results.
Note: The mRNA and vector-based therapies are not really new technologies. MRNA and vector-based therapies have been in use since 2012 to treat patients with cancers, inherited immunodeficiencies, metabolic, eye, neuro-muscular diseases, even hypercholesterolemia.
2. Ivermectin and COVID-19
Even with successful vaccines and other preventable measures in place, the availability of outpatient treatments with solid evidence has a critical role to play in ending this pandemic.
Some doctors and media channels argue that there is very little evidence to support the use of ivermectin to treat COVID-19. However, you can find a summary of more than 60 studies on ivermectin done by more than 500 authors from
c19ivermectin.com (constantly updated).
As of August 2021, there are more than 80 on-going trials globally on Ivermectin for treatment and prevention of COVID-19 on
covid-nma.com.
The Critical Care physicians of the FLCCC Alliance conducted a comprehensive review of the rapidly emerging scientific evidence on Ivermectin from studies conducted around the world. The link to their review that has been published in a peer-reviewed journal is
HERE. This review led the team to develop the
I-Mask+ Protocol and to call for its urgent adoption by health authorities — who could subsequently issue immediate guidance for the nation’s prescribing physicians.
3. Dietary Supplements (Vitamin D, C, Zinc, Quercetin) and COVID-19
Based on the AAPS algorithm or flowchart below, nutraceuticals are the first line treatment for mild COVID-19 patients.
There are more than 70 types of supplements that are being tested for COVID-19. You can review the details of these trials on
clinicaltrials.gov.
Quercetin,
Hydroxychloroquine, EGCG and Zinc are among the handful of COVID-19 treatments that are being studied as potential candidates that might influence the outcome in the prevention and management of COVID-19. Hydroxychloroquine, Quercetin and EGCG (EpiGalloCatechin Gallate) are all zinc ionophores. Meaning they all transport zinc into the cells.
For early outpatient protocol (COVID-19 positive), the Front Line COVID-19 Critical Care Working Group, FLCCC I-MASK+ protocol recommends (updated August 10, 2021):
- Ivermectin: 0.4–0.6 mg/kg per dose (take with or after meals) — one dose daily, take for 5 days or until recovered. (Find a Doctor). Use upper dose range if: 1) in regions with more aggressive variants; 2) treatment started on or after day 5 of symptoms or in pulmonary phase; or 3) multiple comorbidities/risk factors.
- Fluvoxamine: 50 mg twice daily for 10–14 days. Add to ivermectin if: 1) minimal response after 2 days of ivermectin; 2) in regions with more aggressive variants; 3) treatment started on or after day 5 of symptoms or in pulmonary phase; or 4) numerous co-morbidities/risk factors. Avoid if patient is already on an SSRI (selective serotonin reuptake inhibitor).
- Vitamin D3: 4000 IU/day. (Amazon)
- Vitamin C: 500 - 1,000 mg BID (twice daily) (Amazon)
- Quercetin: 250 mg twice a day. (Amazon)
- Melatonin: 10 mg before bedtime (causes drowsiness). (Amazon)
- Zinc: 100 mg/day. Zinc lozenges are preferred. (Amazon)
- Nasal/oral rinse: 3 x daily – gargle (do not swallow) antiseptic mouthwash with cetylpyridinium chloride (e.g. Crest, Scope mouthwash™), ListerineTM with essential oils, or povidone/iodine 1 % solution as alternative (Betadine® Antiseptic Sore Throat Gargle™). (Reference, page 13)
- Aspirin: 325 mg/day unless contraindicated. (Amazon)
- Aspirin: 325 mg/day unless contraindicated. (Amazon)
- Pulse Oximeter: FLCCC also recommend monitoring your oxygen saturation with a pulse oximeter and to go to the hospital if you get below 94%. (Amazon)
A word about quercetin: Some physicians are recommending this supplement to reduce viral illnesses because quercetin acts as a zinc ionophore to improve zinc uptake into cells. It is much less potent than HCQ (hydroxychloroquine) as a zinc transporter, and it does not reach high concentrations in lung cells that HCQ does. Quercetin may help reduce risk of viral illness if you are basically healthy. But it is not potent enough to replace HCQ for treatment of COVID once you have symptoms, and it does not adequately get into lung tissue unless you take massive doses (3-5 grams a day), which cause significant GI (gastrointestinal) side effects such as diarrhea.
Note: The dosages for prevention and treatment protocols are different as the risks and benefits are different for the respective situations. Prevention protocol is for those who are not COVID-19 positive and the treatment protocol is for those who are COVID-19 positive. Do take note that 'early' treatment is important for the best possible outcome.
4. Bamlanivimab plus Etesevimab, Casirivimab plus Imdevimab and Sotrovimab (Monoclonal Antibodies)
The NIH COVID-19 Treatment Guidelines Panel
recommends using one of the following anti-SARS-CoV-2 monoclonal antibodies, listed in alphabetical order, to treat nonhospitalized patients with mild to moderate COVID-19 who are at high risk of clinical progression, as defined by the Emergency Use Authorization (EUA) criteria:
- Bamlanivimab plus etesevimab; or
- Casirivimab plus imdevimab; or
- Sotrovimab.
There is some confusion among members of the public, thinking that they are similar to vaccine. Monoclonal antibody is a 'treatment' for COVID-19, not preventive. Monoclonal antibodies are not the same as the COVID-19 vaccine rolling out right now (refer above).
5. Hydroxychloroquine and COVID-19
We have combined hydroxychloroquine and chloroquine under 1 category.
Hydroxychloroquine, a less toxic derivative of Chloroquine is a widely used medication by people with lupus or arthritis. It was first approved in the 1950s. Chloroquine is used to treat and prevent malaria and amebiasis.
In general, hydroxychloroquine has not been associated with improved survival among hospitalized COVID-19 patients in the majority of observational studies. Do take note that for some of these studies, hydroxychloroquine was given late or was not combined with zinc. However, 'EARLY' treatment with hydroxychloroquine AND 'zinc' shows
promising results instead.
- HCQ (Hydroxychloroquine) is not effective when used very late with high dosages over a long period (RECOVERY/SOLIDARITY), effectiveness improves with earlier usage and improved dosing.
- Early treatment shows positive effects.
- Negative evaluations typically ignore treatment time, often focusing on a subset of late stage studies and did not include zinc.
6. Nasal Sprays and COVID-19
Do nasal sprays actually work against COVID-19?
Below, we've listed the best nasal sprays for COVID-19. Do note that some of them are available as over-the-counter medications but some are still under clinical trial.
1. Nitric Oxide Nasal Spray
2. Iota-Carrageenan Nasal Spray
3. Povidone Iodine Nasal Spray
4. AeroNabs Nasal Spray for COVID
5. Xlear Nasal Spray
6. Taffix Nasal Spray
7. Halberd COVID-19 Preventative Nasal Spray
8. BioBlock®, a Novel Prophylactic Nasal Spray
7. Inhaled Budesonide and COVID-19
In addition to the obvious symptoms that COVID patients get such as fever and cough during the initial viral phase, they may also get symptoms and signs related to two distinct processes i.e. hyperinflammation (with out without cytokine storm) and a hypercoagulable state (hpercoagulability).
In April 2021, The Lancet
reported evidence of UK’s first effective drug to treat COVID-19 in patients at home, inhaled budesonide, showing the treatment can reduce recovery time by a median of three days. The treatment has since been included in clinical guidelines for treating early-stage COVID-19 across the UK, Canada and India.
Budesonide is a steroid sold under the trade name Pulmicort by AstraZeneca Plc and is also used for treating smoker's lung. The
28-day study of 146 patients suggested that those who inhaled budesonide reduced the risk of urgent care or hospitalization by 90 per cent when compared with usual care.
Budesonide as an anti-viral?
Budesonide is widely known as an anti-inflammatory and not anti-viral. However,
this study (Viruses - July 2021) demonstrated a dose-dependent inhibition of SARS-CoV-2 that was comparable between all viral variants tested and could indicate a multimodal mode of action of budesonide against SARS-CoV-2 and COVID-19, which could contribute to an improved clinical performance.
Inhaled corticosteroids (budesonide): Unclear benefit.
Two recent Randomised Controlled Trials (
STOIC 2021,
PRINCIPLE 2021) have demonstrated more rapid symptomatic improvement in ambulatory patients with COVID-19 treated with inhaled budesonide, however, there was no difference in the rate of hospitalization. It should be noted that both these studies were open label (no placebo in the control arm) and that the primary end-point was subjective (time to symptom resolution). Corticosteroids downregulate the expression of interferons (hosts primary antiviral defenses) and downregulated ACE-2 expression (harmful).
Furthermore, two population level studies (
Lancet Resp Med 2021,
OpenSAFELY 2021) suggest that inhaled corticosteroids may increase the risk of death in patients with COVID-19. Based on these data the role of inhaled corticosteroids in the 'early phase' of COVID-19 is unclear.
Clinical Research
There are several open-label studies currently open to recruitment examining the role of inhaled budesonide in COVID-19 infection (ISRCTN86534580,
NCT04355637,
NCT04331054) and others investigating the role of inhaled ciclesonide (
NCT04330586,
NCT04377711,
NCT04381364,
NCT04356495); whether these studies also show an effect on long COVID will be of importance.
Inhaled Budesonide vs Nasal Spray Budesonide
There is also a common confusion between 'inhaled' budesonide and 'nasal spray' budesonide. They are not the same. Nasal sprays are meant for the nose and is commonly used for allergic rhinitis. However, for the drug to get to the lungs, budesonide needs to be in an inhalation device (e.g. Pulmicort Turbohaler).
Note: Inhaled budesonide is part of the AAPS protocol (above) and is recommended when you have breathing or respiratory problems.
8. Povidone Iodine and Mouthwash
Due to the presence of angiotensin-converting enzyme 2 (ACE2) in the oral gingival epithelium and salivary glands, the human oral cavity may act as a reservoir for SARS-CoV-2. The ACE2 present on the host cell membrane acts as the primary entry receptor for SARS-CoV-2. Evidence indicates that the saliva of SARS-CoV-2-infected individuals contains high amounts of viral RNA and that aerosols formed from the saliva can act as a potential vector for viral transmission.
Povidone iodine (PVP-I) is an antiseptic that has been used for over 150 years. It's already proven that different concentration of PVP-I can deactivate COVID-19 virus.
In a primary prevention study in Singapore, a povidone-iodine throat spray administered three times daily proved to be highly effective in reducing the risk of laboratory confirmed SARS-CoV-2 infection.
Antiseptic mouthwashes/throat rinses (chlorhexidine, povidone-iodine, cetylpyridinium chloride) and/or povidone-iodine (Betadine) nasal spray/antiseptic can be applied 2 times per day. A mouth wash containing cetylpyridinium chloride (CPC) has broad antimicrobial properties and has been shown to be effective in controlling gingivitis and gingival plaque.
An in-vitro study demonstrated that CPC was highly viricidal against COVID-19 virus.
Oropharyngeal and nasal sanitization will likely reduce the viral load in the upper airways, thereby reducing the risk of symptomatic disease and likely reducing disease severity. This may be particularly important with the Delta variant which replicates to achieve viral high loads in the nasopharynx/ oropharynx. (Ref, Page 13)
For a summary of 6 studies on povidone-iodine, check out c19pvpi.com (constantly updated).
9. Aspirin, Antiplatelet agents and antithrombotics
In addition to the obvious symptoms that COVID patients get such as fever and cough during the initial viral phase, they may also get symptoms and signs related to two distinct processes i.e. hyperinflammation (with out without cytokine storm) and a hypercoagulable state. A hypercoagulable state is the medical term for a condition in which there is an abnormally increased tendency toward blood clotting (coagulation).
COVID-19 patients have described chest heaviness associated with the possibility of pulmonary thrombosis (Bhandari et al., 2020). Autopsy studies have described pulmonary micro thrombosis and overt embolism with deep venous thrombus found in over half of fatal COVID-19 cases (Ackermann et al., 2020; Burlacu et al., 2020). These observations support the hypothesis that a unique endothelial injury and thrombosis are playing a role in oxygen desaturation, a cardinal reason for hospitalization and supportive care (Zhang et al., 2020b).
Because thromboxane A2 is markedly upregulated with SARS-CoV-2 infection,
early administration of aspirin 325 mg per day is advised for initial antiplatelet and anti-inflammatory effects (Chow et al., 2020; Glatthaar-Saalmüller et al., 2017; Turshudzhyan, 2020; A. Gupta et al., 2020a).
In a retrospective study of 2773 COVID-19 inpatients, 28% received anticoagulant therapy within 2 days of admission, and despite being used in more severe cases, anticoagulant administration was associated with a reduction in mortality, HR = 0.86 per day of therapy, 95% CI: 0.82-0.89; P<< 0.001. Pre-emptive use of low molecular weight heparin or novel anticoagulants have been associated with >> 50% reduction in COVID-19 mortality (Billett et al., 2020).
Finally, many acutely ill outpatients also have general indications or risk for cardioembolic/venous thromboembolic prophylaxis applicable to COVID-19 (Moores et al., 2020; Ruocco et al., 2020). There are ambulatory randomized trials of aspirin and novel oral anticoagulants underway. However, given reports of catastrophic stroke and systemic thromboembolism and the large reductions in mortality for both prophylactic and therapeutic use, administration of aspirin 325 mg po qd for all COVID-19 high-risk patients and systemic anticoagulation is prudent in patients with a history of heart, lung, kidney, or malignant disease (Yamakawa et al., 2020).
10. AzithromycinAzithromycin is a widely prescribed generic antibiotic. While it's mainly used to fight bacteria, not viruses, there is some research suggesting the drug has antiviral properties.
As of August 2021, there are more than 120 studies that have been launched to investigate the benefits of Azithromycin against COVID-19. You can review the status of these trials on
clinicaltrials.gov. Several trials are testing azithromycin in combination with hydroxychloroquine.
In a retrospective cohort study published in
the Lancet, it is found that 50% of COVID-19 patients who died had bacterial co-infections in pneumonia. Bacterial coinfections have also been shown to increase the risk of mortality for COVID-19 patients [
Chen et al].
One potential concern is
serious heart side effects. Both drugs can cause abnormal changes in the rhythm of the heart. These can be fatal, particularly for susceptible patients who already have heart problems. Many studies are using EKG tests to closely monitors patients receiving this treatment combination.
While QT-prolonging medication use has been associated with increased risk of death, this risk may be smaller than the potential benefit from treatment of COVID-19 for some patients (
American College of Cardiology).
In a study on the relationship of
faecal bacteria and COVID-19 virus by Petrillo et al, preliminary results suggest that SARS-CoV-2 replicates in bacterial growth medium inoculated with a stool sample from an infected patient and that the replication follows bacterial growth. Although based on a single observation, their results suggest that the COVID-19 virus or parts thereof, is capable of replicating also outside the human body.
Note: Azithromycin is sold under the trade name Zithromax by Pfizer.
What's New
Check out this review on a highly navigable website that has continuously updated the evidence-based research on early COVID-19 treatments since June 2020:
C19Early.com.
For a dynamic review on COVID-19 Prevention:
Review of COVID-19 Prevention Evidence.






.png)



.png)


Comments
Post a Comment